DNA sequence-dependent ionic currents in ultra-small solid-state nanopores
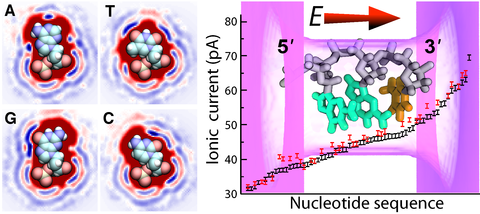
Measurements of ionic currents through nanopores partially blocked by DNA have emerged as a powerful method for characterization of the DNA nucleotide sequence. Although the effect of the nucleotide sequence on the nanopore blockade current has been experimentally demonstrated, prediction and interpretation of such measurements remain a formidable challenge. Using atomic resolution computational approaches, here we show how the sequence, molecular conformation, and pore geometry affect the blockade ionic current in model solid-state nanopores. We demonstrate that the blockade current from a DNA molecule is determined by the chemical identities and conformations of at least three consecutive nucleotides. We find the blockade currents produced by the nucleotide triplets to vary considerably with their nucleotide sequence despite having nearly identical molecular conformations. Encouragingly, we find blockade current differences as large as 25% for single-base substitutions in ultra small (1.6 nm x 1.1 nm cross section; 2 nm length) solid-state nanopores. Despite the complex dependence of the blockade current on the sequence and conformation of the DNA triplets, we find that, under many conditions, the number of thymine bases is positively correlated with the current, whereas the number of purine bases and the presence of both purine and pyrimidines in the triplet are negatively correlated with the current. Based on these observations, we construct a simple theoretical model that relates the ion current to the base content of a solid-state nanopore. Furthermore, we show that compact conformations of DNA in narrow pores provide the greatest signal-to-noise ratio for single base detection, whereas reduction of the nanopore length increases the ionic current noise. Thus, the sequence dependence of nanopore blockade current can be theoretically rationalized, although the predictions will likely need to be customized for each nanopore type.